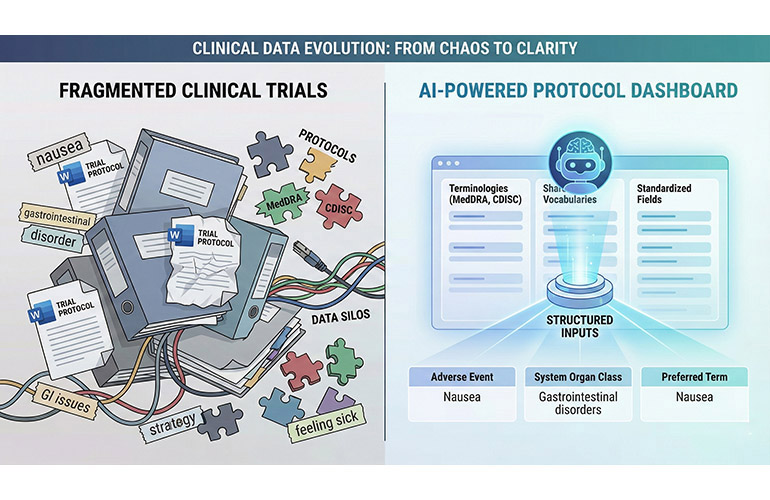
 In June 2025, the Food and Drug Administration rolled out Elsa, an agency-wide generative AI assistant that officials say is already helping to speed the review of new drugs and devices and shrink weeks of paperwork into minutes. It is a vision that could reshape how lifesaving therapies reach patients, but Elsa’s first six months have exposed growing pains: internal pushback, documented hallucinations, and a more fundamental problem regulators have long overlooked: fragmented, disparate,…
In June 2025, the Food and Drug Administration rolled out Elsa, an agency-wide generative AI assistant that officials say is already helping to speed the review of new drugs and devices and shrink weeks of paperwork into minutes. It is a vision that could reshape how lifesaving therapies reach patients, but Elsa’s first six months have exposed growing pains: internal pushback, documented hallucinations, and a more fundamental problem regulators have long overlooked: fragmented, disparate,…



