FOR IMMEDIATE RELEASE
March 2026 | Source: Market Research Future (MRFR)
Propelled by Rising GI Disorder Prevalence, Superior Clinical Profile over PPIs, Expanding Generic Pipeline, and Accelerating Global Regulatory Approvals
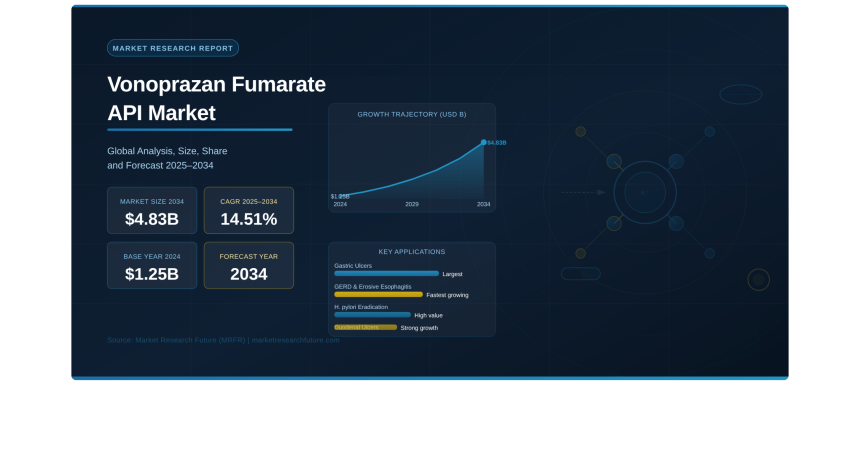
Pune, India — The global Vonoprazan Fumarate API Market is emerging as one of the most compelling growth stories in the active pharmaceutical ingredient (API) landscape, according to the latest Vonoprazan Fumarate API Market report published by Market Research Future (MRFR). Valued at USD 1.25 billion in 2024, the market is projected to grow from USD 1.43 billion in 2025 to USD 4.83 billion by 2034, representing a compound annual growth rate (CAGR) of 14.51% during the forecast period 2025 to 2034. This extraordinary trajectory positions Vonoprazan Fumarate as one of the most transformative molecules in modern gastroenterology, rapidly displacing conventional proton pump inhibitors (PPIs) as the acid suppression treatment of choice for clinicians and patients worldwide. The Vonoprazan Fumarate API Market growth is deeply aligned with broader Longevity Market size trends, as aging global populations increasingly drive demand for effective, fast-acting gastrointestinal therapies that address the rising prevalence of acid-related disorders across all demographic cohorts.
Vonoprazan Fumarate API Market Summary
Vonoprazan Fumarate is a next-generation, orally active potassium-competitive acid blocker (P-CAB) that represents a fundamental pharmacological advancement over conventional proton pump inhibitors. Unlike PPIs, which require activation in an acidic environment and are subject to variable metabolism through the CYP2C19 enzyme pathway, Vonoprazan operates through a potassium-competitive mechanism that delivers rapid, potent, and durable acid suppression from the very first dose — regardless of meal timing, patient genetic polymorphism, or pH conditions. This superior pharmacological profile has made Vonoprazan Fumarate the API of choice for treating gastroesophageal reflux disease (GERD), gastric ulcers, duodenal ulcers, Helicobacter pylori eradication regimens, and erosive esophagitis.
According to MRFR analysis, the Vonoprazan Fumarate API Market size reached USD 1.25 billion in 2024 and is forecast to reach USD 4.83 billion by 2034 at a 14.51% CAGR. The market encompasses both branded and generic API formulations, spanning oral tablets, oral suspensions, and injectable dosage forms across pharmaceutical manufacturers, contract manufacturers, API suppliers, and B2B licensing partners globally. The Vonoprazan Fumarate API Market share is distributed across North America, Asia-Pacific, Europe, South America, and the Middle East and Africa, with each region exhibiting distinct adoption patterns driven by regulatory landscapes, physician preferences, and healthcare system infrastructure.
Request Your Sample Copy of This Strategic Report:
https://www.marketresearchfuture.com/sample_request/30898
Vonoprazan Fumarate API Market Growth Factors
The Vonoprazan Fumarate API Market growth is driven by a convergence of clinical, regulatory, demographic, and commercial forces that are accelerating global adoption at an exceptional pace:
- Rising Global Burden of Acid-Related Gastrointestinal Disorders: Gastroesophageal reflux disease affects approximately 18 to 27 percent of adults in Western countries, while gastric and duodenal ulcers, H. pylori infection, and erosive esophagitis collectively impact hundreds of millions of patients worldwide. The increasing prevalence of these conditions — driven by dietary shifts, obesity epidemics, sedentary lifestyles, stress, and aging populations — is creating a structurally expanding patient pool requiring effective, sustained acid suppression therapy.
- Superior Clinical Profile Over Traditional PPIs: Vonoprazan’s potassium-competitive mechanism delivers faster onset of action, more consistent 24-hour acid control, and superior H. pylori eradication rates compared to conventional PPIs such as omeprazole, lansoprazole, and esomeprazole. Clinical trial data demonstrate that Vonoprazan achieves mucosal healing and symptom relief at significantly higher rates — driving accelerating physician preference switching across key markets and contributing directly to Vonoprazan Fumarate API Market expansion.
- Accelerating Regulatory Approvals and Global Market Access: Vonoprazan has received regulatory approval in Japan, the United States (via FDA approval for GERD and H. pylori eradication), and increasingly in European and Asian markets. Each new regulatory approval unlocks a new national market for branded and generic API supply, creating compounding demand across the forecast period and establishing Vonoprazan as a truly global therapeutic standard.
- Expanding Generic Pharmaceutical Pipeline: As branded exclusivity windows narrow in key markets, generic pharmaceutical manufacturers — particularly in India, China, and South Korea — are investing heavily in Vonoprazan Fumarate API synthesis, purity optimization, and Drug Master File (DMF) submissions. This generic entry is expected to dramatically expand market volume by improving affordability and accessibility, particularly across price-sensitive emerging markets in Asia-Pacific, Latin America, and the Middle East.
- Strategic Collaborations, Licensing, and B2B API Partnerships: Leading API manufacturers are entering into supply agreements, co-development partnerships, and licensing arrangements to accelerate global distribution of high-purity Vonoprazan Fumarate API. These B2B ecosystems are strengthening supply chain resilience and enabling formulators to bring finished dosage forms to market faster across regulated and semi-regulated markets worldwide.
🛒 Buy Now Premium Research Report –
https://www.marketresearchfuture.com/checkout?currency=one_user-USD&report_id=30898
Vonoprazan Fumarate API Market Trends
The Vonoprazan Fumarate API Market is being shaped by a set of defining clinical, manufacturing, and commercial trends that are determining competitive positioning through 2034:
- Generic Market Explosion and API Commoditization: The expiry of Vonoprazan’s branded exclusivity in key markets is triggering a wave of generic API manufacturer entries, led by Indian and Chinese pharmaceutical companies scaling high-purity synthesis capabilities. This trend is expected to significantly reduce API pricing over the medium term while dramatically expanding market volume — a dynamic that closely mirrors generic PPI market development patterns observed in previous decades.
- Purity Grade Innovation — Above 99% Segment Leadership: The Above 99% purity segment holds the largest share of the Vonoprazan Fumarate API Market, accounting for approximately 60% of revenue, as pharmaceutical formulators and regulatory agencies increasingly mandate ultra-high purity specifications for both branded and generic finished dosage form production. Investment in purity optimization and advanced synthesis processes is a key competitive differentiator among leading API manufacturers.
- Injectable and Oral Suspension Format Diversification: While oral tablets dominate the current market — accounting for approximately 80% of dosage form volume — injectable Vonoprazan formulations are gaining traction in hospital and acute care settings for patients with severe gastrointestinal conditions requiring rapid parenteral intervention. Oral suspension formats are also emerging as an important growth segment for pediatric and dysphagia patient populations, expanding the addressable market beyond the traditional adult oral solid dosage segment.
- Combination Therapy Innovation: Vonoprazan is increasingly being formulated as part of combination H. pylori eradication regimens — paired with amoxicillin and clarithromycin — following landmark clinical data demonstrating superior eradication rates compared to PPI-based triple therapy. These combination products represent a significant market opportunity for API manufacturers supplying multi-drug formulation pipelines.
- Patient-Centric Care and Personalized Medicine Integration: The broader shift toward personalized medicine is creating demand for tailored Vonoprazan treatment protocols that account for individual patient GI physiology, disease severity, and comorbidities. Digital health platforms and telehealth services are also improving patient access to Vonoprazan-based therapies in markets with historically limited gastroenterology specialist density.
Vonoprazan Fumarate API Market Segment Analysis
The Vonoprazan Fumarate API Market share is distributed across three primary segmentation dimensions — dosage form, application, and type — each reflecting distinct clinical demand characteristics and manufacturing economics:
By Dosage Form, the Oral segment dominates the Vonoprazan Fumarate API Market, driven by the convenience, patient compliance advantages, and well-established oral bioavailability profile of Vonoprazan tablets. The oral tablet format has become the cornerstone of first-line acid suppression therapy in both outpatient and primary care settings globally, and its dominance is expected to persist through 2034. The Injectable segment, while currently representing a smaller market share, is growing at an accelerating rate driven by hospital formularies adopting parenteral Vonoprazan for acute GI bleeding, post-surgical acid prophylaxis, and intensive care applications. Oral Suspension formats represent the fastest-growing dosage form innovation pipeline, capturing emerging opportunities in pediatric gastroenterology and adult dysphagia management.
By Application, Gastric Ulcers represent the single largest treatment application, driven by the high global prevalence of this condition and Vonoprazan’s demonstrated superiority in achieving faster ulcer healing and lower recurrence rates versus PPIs. Duodenal Ulcers are the second-largest application segment, with growing clinical adoption supported by strong comparative efficacy data. GERD and Erosive Esophagitis are the fastest-growing application segments, as physicians increasingly recognize Vonoprazan’s advantages in delivering sustained nighttime acid control and superior mucosal healing — areas where conventional PPIs have historically underperformed. H. pylori Eradication is emerging as a uniquely high-value application segment following landmark global clinical trials confirming Vonoprazan-based triple therapy superiority.
By Type, the Branded segment maintains strong clinical and institutional trust, particularly in developed markets where Takeda’s branded Vonoprazan products carry premium positioning and physician familiarity. The Generic segment is the faster-growing type category, driven by increasing API manufacturing capacity in India, China, and South Korea, expanding DMF filing activity, and the market access imperative in emerging economies where branded pricing creates significant affordability barriers.
Vonoprazan Fumarate API Market Regional Analysis
The Vonoprazan Fumarate API Market regional landscape reflects Japan’s pioneering role, North America’s recent acceleration, Europe’s steady expansion, and Asia-Pacific’s emergence as the fastest-growing region — a distribution that connects meaningfully to the Longevity Market share pattern, where aging demographics are the primary driver of therapeutic demand across all regions:
North America leads the global Vonoprazan Fumarate API Market in revenue terms, accounting for approximately 35 to 40 percent of total market value. The United States represents the single largest national market following the FDA’s approval of Vonoprazan for GERD treatment and H. pylori eradication — approvals that have catalyzed rapid uptake across the US gastroenterology and primary care physician community. The well-developed US pharmaceutical supply chain, strong payer reimbursement frameworks, and high awareness of Vonoprazan’s clinical advantages are all sustaining North America’s dominant regional position through the forecast period.
Asia-Pacific is the fastest-growing regional market in the Vonoprazan Fumarate API Market and is expected to reach approximately USD 1.5 billion by 2025, reflecting the region’s exceptional growth momentum. Japan holds a unique historical position as Vonoprazan’s country of origin and primary clinical development market, with Takeda’s original branded product — marketed as Takecab — having established deep physician and formulary penetration. China, India, South Korea, and Australia are experiencing rapid market expansion as regulatory approvals broaden, local generic API manufacturing scales, and awareness of Vonoprazan’s superior clinical profile spreads across the region’s vast gastroenterology patient population. The demographic parallel with the Longevity Market growth trajectory in Asia-Pacific is striking — both markets are being simultaneously driven by aging populations, rising gastrointestinal disease prevalence, and substantial healthcare infrastructure investment.
Europe maintains steady and growing demand for Vonoprazan Fumarate API, fueled by guideline inclusion in major national gastroenterology protocols, increasing physician preference for Vonoprazan over legacy PPIs particularly for H. pylori eradication, and expanding pharmaceutical distribution networks across Germany, France, the United Kingdom, Italy, Spain, and the Nordic countries. The European Medicines Agency (EMA) regulatory framework is progressively supporting broader market access for both branded and generic Vonoprazan formulations.
South America and the Middle East and Africa represent emerging Vonoprazan Fumarate API Markets, showing consistent growth momentum as healthcare access improves, generic API versions become available at accessible price points, and the prevalence of H. pylori infection — particularly elevated across both regions — drives robust clinical demand for effective eradication therapies.
Key Market Players
The Vonoprazan Fumarate API Market is led by a competitive ecosystem of branded pharmaceutical innovators, specialized API manufacturers, and generic producers scaling global supply capacity. Key players identified in this Vonoprazan Fumarate API Market report include Takeda Pharmaceutical Company Ltd. (Japan) — the pioneering developer and branded market leader — Cisen Pharmaceutical Co. Ltd. (China), Ami Lifesciences Private Limited (India), Dr. Reddy’s Laboratories Ltd. (India), Hetero Drugs Ltd. (India), Metrochem API Private Limited (India), Virupaksha Organics Ltd. (India), Zhejiang Hengkang Pharmaceutical Co. Ltd. (China), Maithri Drugs Pvt. Ltd. (India), Nippon Zhenlei Chemical (Japan), Xian Wanlong Pharmaceutical (China), HONOUR LAB LTD (India), Nifty Labs (India), and Optimus Pharma (India). These companies are collectively investing in GMP-certified manufacturing capacity expansion, high-purity synthesis optimization, DMF filing programs, and strategic licensing partnerships to capture the market’s extraordinary 14.51% CAGR growth opportunity through 2034.
About Market Research Future (MRFR)
Market Research Future (MRFR) is a leading global market research organization providing comprehensive, data-driven analysis across a wide spectrum of industries. For full report details, visit: www.marketresearchfuture.com/reports/vonoprazan-fumarate-api-market-30898
Contact: info@marketresearchfuture.com | +1 (855) 661-4441